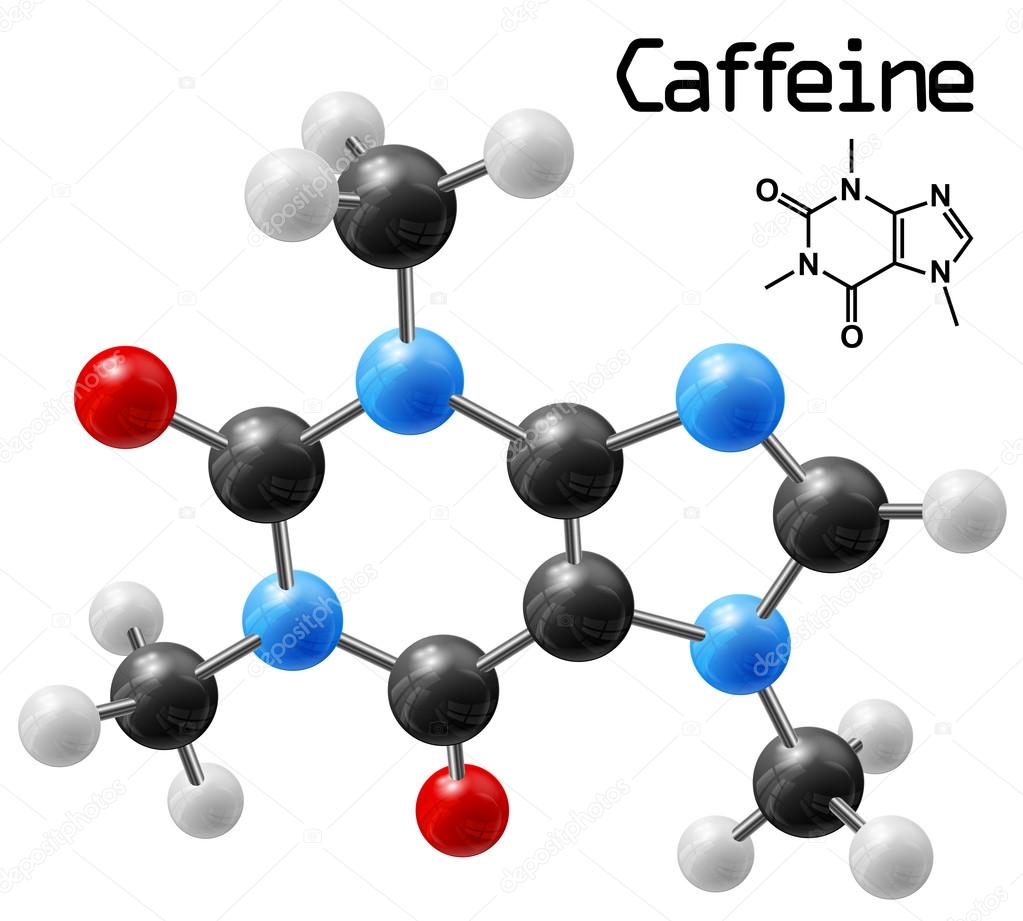
There are several online databases that provide estimates of caffeine content of certain foods and beverages such as coffee and tea. Consumers should take care when consuming for the first time a new packaged food containing added caffeine if the amount of caffeine in the food is not declared on the label. Many packaged foods, including beverages and dietary supplements containing caffeine, voluntarily provide information on the label as to how much caffeine they contain. How do you know how much caffeine a food or beverage contains? It’s also found in some plants used as flavorings, such as guarana, or alternative teas popular in South American, such as yerba mate (Ilex paraguariensis) and Ilex guayusa.Ĭaffeine may also be added as an ingredient to foods and beverages. Which kinds of foods and beverages contain caffeine?Ĭaffeine can be found naturally in the plants we use to make coffee, tea and chocolate. * Sports Drinks and Energy Drinks for Children and Adolescents: Are They Appropriate? American Academy of Pediatrics June 2011 1. The American Academy of Pediatrics has stated that “there is heightened awareness of the risks of caffeine use, abuse, and even toxicity in children and adolescents.”* Consult your health care provider for advice regarding your child’s caffeine consumption. You should learn to recognize any of the possible Lewis structures.Is it okay for kids to consume energy drinks that contain caffeine?Įnergy drinks can have more than twice the caffeine in an equal amount of coffee. While the most complete structure is more useful for the novice chemist, the simplest is quicker to draw and still conveys the same information for the experienced chemist. Why are there different ways for the "same" Lewis structure? It depends what you want to show. It is also common to show only the net charge on the ion rather than all of the formal charges, i.e. Although it is most common to use a line to indicate a bonding pair of electrons they can be shown as electrons, see the left most image below. You have determined the "best" Lewis structure (octets completed and lowest formal charges) for NO 3 -, but there are a number of ways to show this structure.In our present example N can not expand beyond 8 electrons so retains a formal charge of +1, but the S atom below can expand its octet. Note: the octet can be expanded beyond 8 electrons but only for atoms in period 3 or below in the periodic table. Charges of -1 and +1 on adjacent atoms can usually be removed by using a lone pair of electrons from the -1 atom to form a double (or triple) bond to the atom with the +1 charge.the formal charges should match the electronegativity of the atom, that is negative charges should be on the more electronegative atoms and positive charges on the least electronegative atoms if possible. 
formal charges of 0 for as many of the atoms in a structure as possible.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
